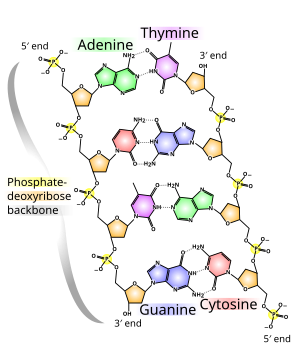
- Living organisms must continually capture, store and use energy, which is the ability to do work.
- The sum of all anabolic and catabolic processes in a cell or organism is called metabolism.
- Work is done when one object applies a force on another object and changes its position or state in motion.
- All forms of energy can be classified as either kinetic energy, which is the energy possessed by all moving objects and can turn into heat energy (movement of particles), mechanical energy (coordinated motions of particles), electromagnetic energy (motion of light) and electrical energy (motion of charged particles).
- Potential energy is stored energy. Created when position changes within an attractive or repulsive force; comes in two different forms, gravitational potential energy (attraction of electrons to protons in a chemical bond) and chemical potential energy (the attraction of electron to protons in a chemical bond)
- Bond energy is the minimum energy required to break one mole of bonds between two species of atoms; a measure of the stability in a chemical bond.
- Activation Energy is the difference between the potential energy level of te transition state and the potential energy of reacting molecules.
- In a chemical reaction, a temporary condition in which the bonds within reactants are breaking and the bonds between products are forming is called the transition state.
- Entropy: a measure of the randomnesss or disorder in a collection of objects or energy
- Free energy: energy that can do useful work.
- Exergonic reaction: a chemical reaction in which the energy of the product is less than the enrgy of the reactants.
- Endergonic reaction: a chemical reaction in which the energy of the products is more that the enrgy of the reactants.
- Oxidation: a chemical reaction in which an atom loses one or more electrons
- Redox reaction: a chemical reaction involving the transfer of one or more electrons from one atom to another
- Reducing agent: a substance that loses an electron in a redox reaction.
- Oxidixing agent: a substance taht gains an electron in a redox reaction.
- Substrate: the reactant that an enzyme acts on when it catalzyes a chemical reaction.
- Active site: the location where the substrate binds to a material
- Competitive inhibitors: substances that complete with the substrate for an enzyme's active site.
- Non competitive inhibitors: substances that attach to a binding site on an enzyme on an enzyme other than the active site.

^CAN YOU FEEL THE ENERGY?!?!?! IT'S OVER 9000!!!!!